Roseway Labs and Mast Cell Action Unite to Advance MCAS Education
Leading UK compounding pharmacy Roseway Labs has officially announced a strategic partnership with Mast Cell Action, a UK-based…

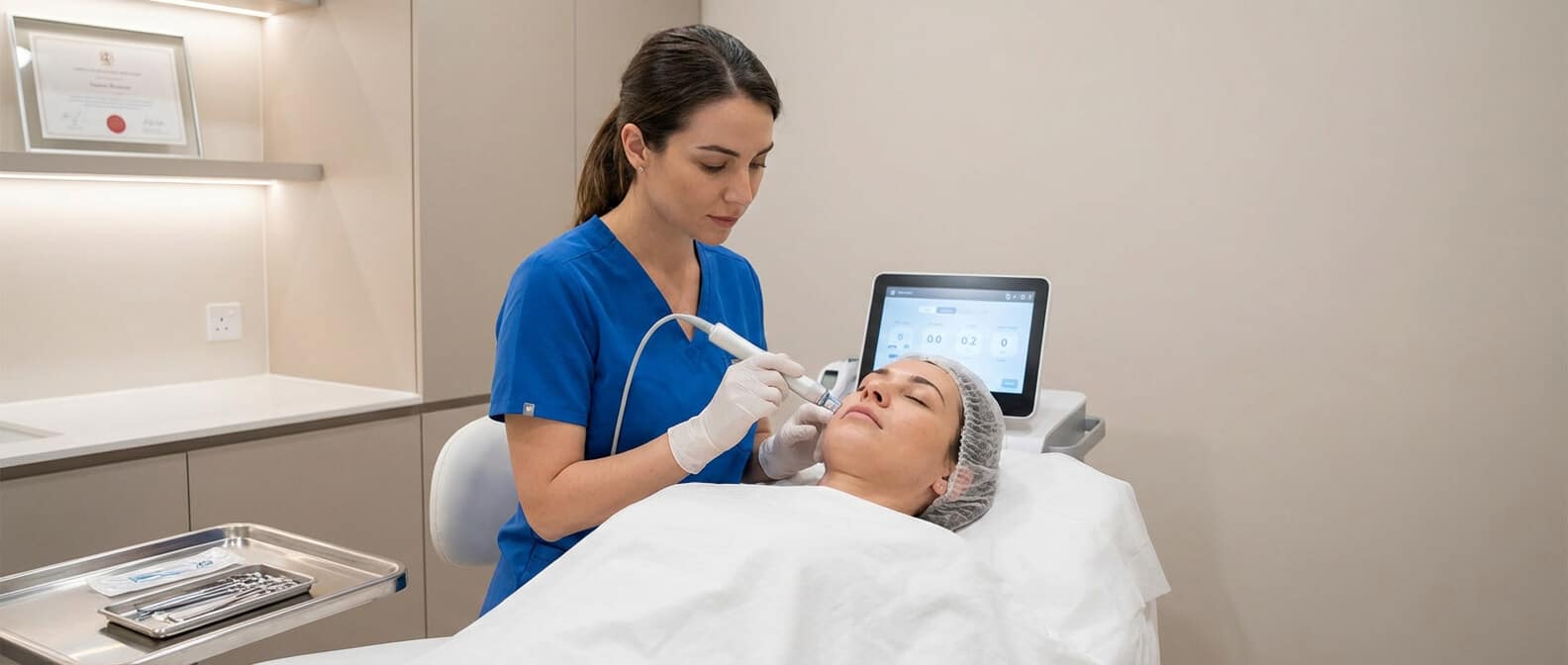
Regenerative medicine is now a mainstream tool in aesthetics, but success depends less on hype and more on control. For clinics and patients, the difference between an inconsistent one-off result and a predictable, repeatable treatment programme lies in the systems behind the syringe. A CE-marked, closed platform such as RegenLab gives clinics that control.
RegenLab’s platform produces three clinically useful outputs from one system, PRP, PRF (ATS) and Cellular Matrix (PRP + hyaluronic acid) so clinics can offer a broad menu of treatments without juggling multiple
suppliers or unstandardised “home-brew” techniques.
The system is a closed, patented technology that uses certified RegenKit® tubes to deliver standardised PRP with consistent platelet yield and sterility, by using a patented gel technology for blood separation. That versatility allows clinicians to offer a broad menu of treatments (scalp and skin PRP, PRF protocols, and Cellular Matrix which combines platelet biology with hyaluronic acid).
The regulatory and practical case for CE marking is simple. A CE mark (and MDR conformity where relevant) means the device and disposables have been manufactured and tested to recognise safety and quality standards, in short, you’re buying a validated, documented process, not just plastic tubes. Clinics increasingly ask for MDR/CE certificates for reassurance.
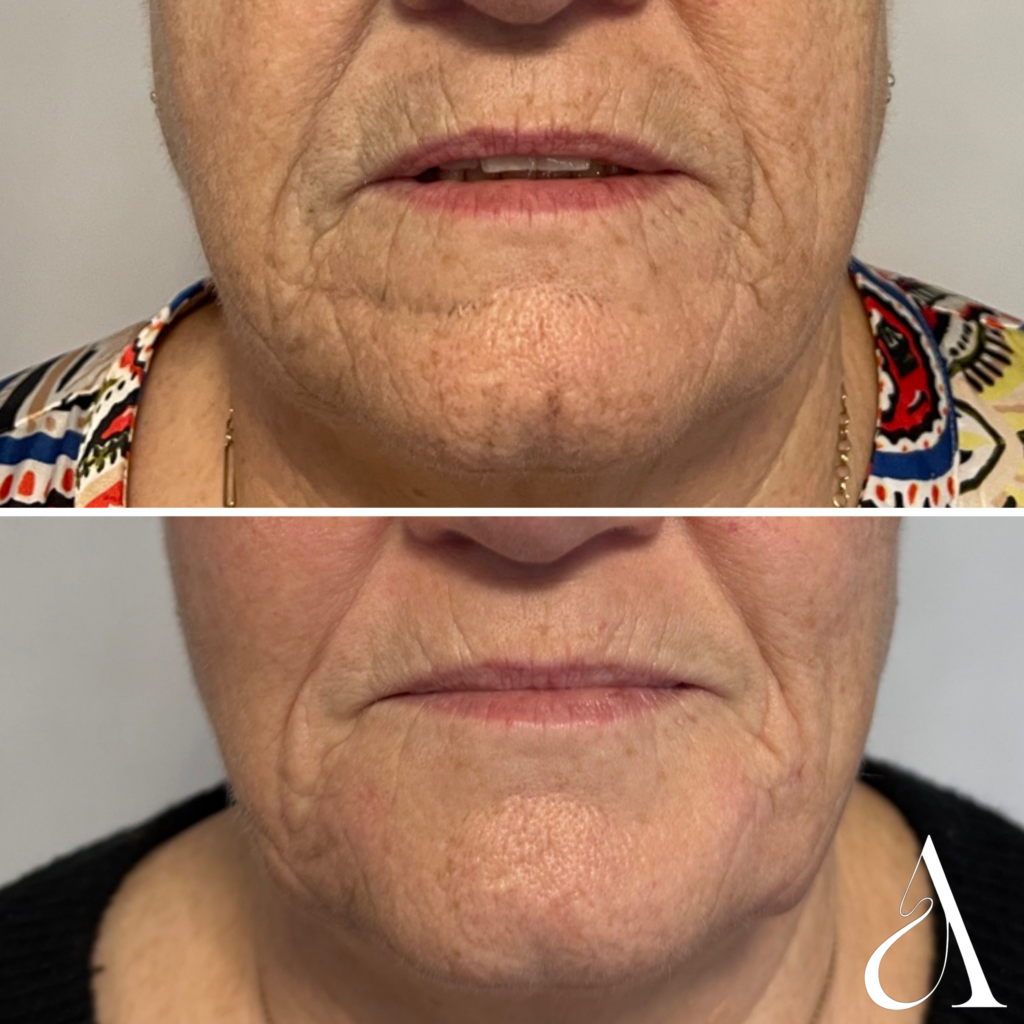
A Aesthetics – Northcoast – patient
had 3 treatments of Cellular Matrix,
30 days apart.
Practical tip for clinics
Always check the manufacturer’s Instructions for Use (IFU). The IFU will state whether the tubes are CE-marked and specifically approved for producing PRP. Some cheaper lab/IVD tubes carry a CE mark for diagnostic use (i.e., they’re certified for putting blood into the tube for testing) but are not validated for PRP preparation and/or they are not certified for taking a processed product back out for use on a patient. Using those tubes can give variable platelet yields, remove proper traceability and conformity, and compromise the documented safety/quality you expect from a regulated PRP system. Always check the IFU and ask the manufacturer for their CE/MDR certificate.
Clinical flexibility without complexity
With RegenPRP, PRF and Cellular Matrix all available within one platform, practitioners can combine biologics and add complementary modalities (for example PRP vs. PRP+ATS/PRF for skin rejuvenation, post laser use of PRP, Cellular Matrix with SkinPen) using standardised preparation steps rather than ad-hoc mixes. That versatility helps clinics tailor treatments to individual patients and to market a clear, evidence-based service offering.
Platform20, an Irish business, based in Duleek, Co. Meath, is the exclusive distributor of RegenLab in Ireland. Both founders have backgrounds in medical device sales, they understand the importance of clinically effective, regulated products and provide local training
and support to help clinics implement validated protocols safely.
Why this matters for Irish clinics.
From a business and governance perspective, a CE-marked, single-
platform solution reduces procurement complexity and makes it easier to train staff, document care, and deliver consistent patient experience. Platform20’s experience bringing RegenLab into Ireland, includes delivery of equipment, clinical training and KOL-led education demonstrating how the right supplier relationship complements a regulated system. Clinicians benefit not only from access to the technology itself, but also from validated protocols and local training to ensure it is used safely and effectively.
A short practical checklist for clinics
In summary, the future of aesthetic regenerative treatments isn’t about the flashiest protocol, it’s about reproducible biology delivered safely. CE-marked platforms such as RegenLab give Irish practitioners a robust, three-in-one foundation, reproducible outputs, regulatory confidence and clinical versatility that helps turn regenerative medicine from an experiment into a dependable service offering.
Gain full access to peer-reviewed protocols, our extensive digital archive, and the Tri-annual print edition delivered to your clinic.
£
250
Per Year
Subscribe
Auto-renews annually. Cancel anytime.
Subscribe to our newsletter for the latest insights from the aesthetics industry.
Subscribe Now