Roseway Labs and Mast Cell Action Unite to Advance MCAS Education
Leading UK compounding pharmacy Roseway Labs has officially announced a strategic partnership with Mast Cell Action, a UK-based…

Non-surgical facial rejuvenation has progressed significantly beyond the paradigm of simple volumetric replacement. Although hyaluronic acid (HA) fillers remain a cornerstone of aesthetic practice, increasing emphasis is now placed on biostimulatory and regenerative injectable therapies that aim to restore dermal architecture, improve tissue quality, and stimulate endogenous collagen production.¹²
This shift reflects evolving patient expectations toward more natural, progressive, and durable outcomes, as well as growing interest in concepts such as collagen preservation and “collagen banking.”³
In response, a diverse range of injectable products has emerged, spanning pure collagen stimulators, dual-action volumisers, and hybrid formulations.
Among these, poly-L-lactic acid (PLLA), calcium hydroxylapatite (CaHA), and poly-D,L-lactic acid (PDLLA)-based injectables, both alone and in combination with HA, have become widely adopted. This review examines four commonly used agents—Sculptra, Radiesse, Juvelook, and Lenisna—focusing on their composition, mechanism of action, duration of effect, delivery techniques, clinical indications, and complication profiles.
Sculptra: A Poly-L-Lactic Acid Biostimulator
Composition
Sculptra is composed of microparticles of poly-L-lactic acid (PLLA), a biocompatible and biodegradable synthetic polymer belonging to the alpha-hydroxy acid family. PLLA has been extensively used in absorbable medical devices and sutures, supporting its established safety profile.⁴⁵
Mechanism of Action
Sculptra acts as a delayed-onset injectable implant. Following injection, PLLA microparticles elicit a controlled subclinical inflammatory response, leading to fibroblast activation and subsequent neocollagenesis.
Newly synthesised collagen progressively replaces the resorbing polymer, resulting in gradual volume restoration and dermal thickening.⁶³
Duration of Action
Clinical improvement develops over several months, with outcomes typically persisting for up to 24 months or longer, depending on treatment protocol and individual patient response.⁴
Method of Delivery
Sculptra is most commonly administered using a cannula and deposited in the deep dermal or subcutaneous plane. Treatment protocols usually involve multiple sessions spaced 4–6 weeks apart, following appropriate reconstitution.³
Areas of Use
Complications
Transient adverse effects include oedema, ecchymosis, erythema, and tenderness. Delayed subcutaneous nodules or granulomas have been reported, particularly with superficial placement or inadequate dilution.
As PLLA is non-reversible, vascular occlusion—though rare—represents a significant complication requiring urgent management.⁷
Radiesse: Calcium Hydroxylapatite Structural Biostimulation
Composition
Radiesse consists of calcium hydroxylapatite (CaHA) microspheres (approximately 30%) suspended in a carboxymethylcellulose gel carrier. CaHA is a naturally occurring mineral component of bone and teeth, conferring excellent biocompatibility.⁸
Mechanism of Action
The gel carrier provides immediate volumisation, while CaHA microspheres act as a scaffold that promotes fibroblast-mediated production of collagen and elastin as the carrier resorbs.⁹
Duration of Action
Clinical effects generally persist for 12–18 months, with variability related to anatomical location and injection technique.⁸¹⁰
Method of Delivery
Radiesse is delivered via needle or cannula into subcutaneous or periosteal planes. Lidocaine-containing formulations are frequently used to enhance patient comfort.⁹
Areas of Use
It is not recommended for the lips or periocular region.¹⁰
Complications
Common reactions include bruising, swelling, and tenderness. Palpable nodules may occur with superficial placement. As CaHA is non-dissolvable, vascular compromise constitutes a serious adverse event requiring prompt intervention.⁷
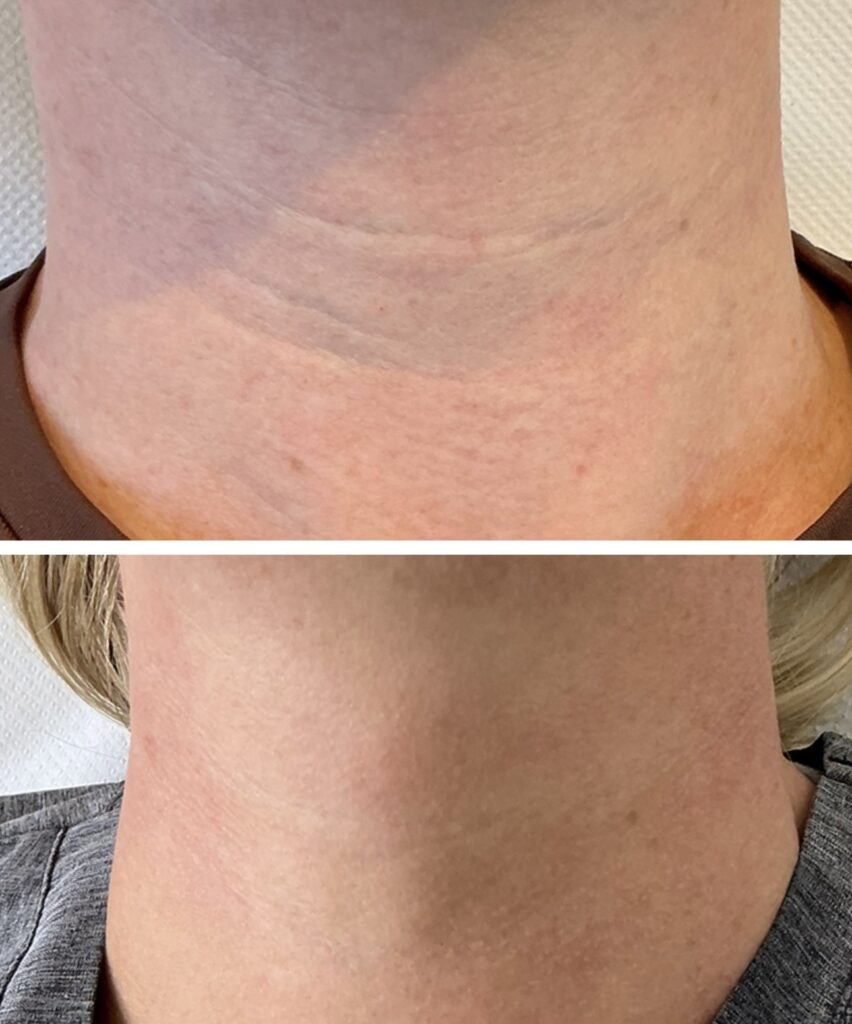
Juvelook: Hybrid PDLLA and Hyaluronic Acid Injectable
Composition
Juvelook is a hybrid injectable comprising poly-D,L-lactic acid (PDLLA) microparticles combined with non-cross-linked hyaluronic acid (HA). PDLLA serves as the biostimulatory component, while HA provides immediate hydration and subtle volumisation.¹¹
Mechanism of Action
Juvelook exhibits a dual mechanism: immediate improvement in hydration and skin luminosity from HA, followed by progressive collagen and elastin synthesis stimulated by PDLLA microparticles. The randomised D- and L-lactic acid polymer structure maximises tissue surface contact. In addition, PDLLA has been shown to modulate the NF-κB inflammatory pathway, reducing collagen degradation and oxidative stress.²
Duration of Action
Immediate effects are observed post-treatment, with progressive improvement over weeks. Clinical benefits typically persist for 12–18 months, with ongoing collagen stimulation beyond visible effects.¹¹
Method of Delivery
Juvelook is injected into the dermis or mid-dermis using a fine needle or cannula. Owing to its small particle size, it may also be administered via microneedling.²
Areas of Use
It also improves pigmentation, pore size, elasticity, and overall skin quality.¹¹
Complications
Transient swelling and bruising are most commonly reported. Due to the small particle size (20–40 μm), vascular occlusion has not been reported. Superficial placement may result in palpable irregularities. Juvelook is thermally modifiable, allowing softening with heat and saline if required.¹²
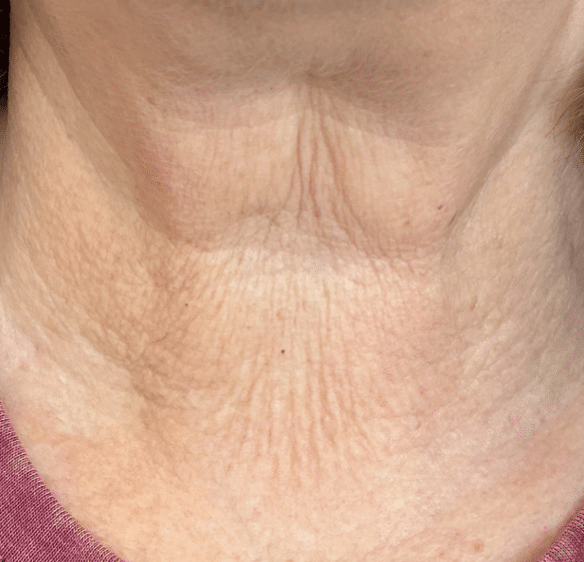
Before Treatment
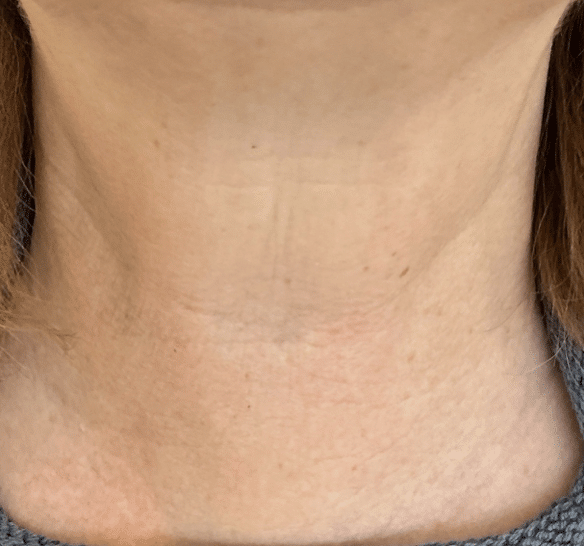
After Treatment
Lenisna: High-Concentration PDLLA Structural Biostimulator
Composition
Lenisna shares the same manufacturer as Juvelook and combines PDLLA microparticles with non-cross-linked HA, but at a substantially higher PDLLA concentration (170 mg versus 42.5 mg). This formulation is designed to address deeper structural volume loss.¹²
Mechanism of Action
Lenisna provides immediate hydration from HA, followed by progressive collagen induction and tissue reinforcement as PDLLA stimulates fibroblasts within deeper tissue planes.²
Duration of Action
Clinical effects typically persist for 18–24 months. Initial protocols often involve 2–3 sessions spaced approximately 8 weeks apart, with maintenance treatment at 18–24 months.¹²
Method of Delivery
Lenisna is delivered using a 25G cannula into the subcutaneous plane to achieve optimal structural support.
Areas of Use
Lenisna is not indicated for periocular use.
Complications
Adverse effects are generally mild and transient, including bruising and swelling. Nodularity may occur with superficial placement. Similar to Juvelook, Lenisna is heat-reversible, allowing corrective softening when required.¹²
Conclusion
Sculptra, Radiesse, Juvelook, and Lenisna represent distinct yet complementary tools within regenerative aesthetic medicine. Understanding their compositional differences, mechanisms of action, and safety profiles allows clinicians to tailor treatments according to anatomical requirements, ageing patterns, and patient expectations.
As biostimulatory injectables continue to evolve, careful patient selection, anatomical expertise, and adherence to evidence-based protocols remain essential to achieving safe, natural, and durable outcomes.
Conflict of Interest Statement
The author declares a professional relationship relevant to the content of this manuscript. The author works as a clinical trainer for FILLMED, a company that has entered into a strategic partnership with VAIM Global, the manufacturer of Lenisna®, Juvelook®, and NCTF®. This role includes educational and training activities related to aesthetic injectable products. The content of this article has been prepared to provide an objective, evidence-based review of biostimulatory injectable treatments. No financial incentives, sponsorship, or editorial influence from the aforementioned companies were received specifically for the preparation of this manuscript.
CaHA = calcium hydroxylapatite
References (Vancouver Style)
Gain full access to peer-reviewed protocols, our extensive digital archive, and the Tri-annual print edition delivered to your clinic.
£
250
Per Year
Subscribe
Auto-renews annually. Cancel anytime.
Subscribe to our newsletter for the latest insights from the aesthetics industry.
Subscribe Now